Piperaquine
Piperaquine | CAS NO: 102458-48-5 is an antimalarial drug primarily used in the treatment of malaria. It is often combined with other medications to enhance its effectiveness and reduce the risk of resistance. Treatment of Malaria: Piperaquine is used in combination therapies to treat malaria caused by Plasmodium falciparum, the parasite responsible for the most severe form of the disease. It is also effective against Plasmodium vivax to some extent.Combination Therapy: Piperaquine is commonly used in combination with artemisinin derivatives, such as artemether or artesunate, to form artemisinin-based combination therapies (ACTs). This combination improves efficacy, reduces the likelihood of resistance, and shortens the duration of treatment.Post-Treatment Prophylaxis: In some cases, piperaquine may be used in combination with other drugs to prevent relapse or re-infection, especially in regions with high malaria transmission rates.
We are the Synchemia Research Chemical specializes in manufacturing and supplying of Piperaquine impurities that meet specific pharmacopeial standards (EP/BP/USP/IP) as well as process-related impurities Piperaquine is an antimalarial drug primarily used in the treatment of malaria. It is often combined with other medications to enhance its effectiveness and reduce the risk of resistance. piperaquine is a valuable component in the fight against malaria, particularly when used in combination with other effective antimalarial drugs. we the Synchemia are ensuring the purity and quality of its impurities which is crucial for pharmaceutical API manufacturing companies for regulatory compliance. Buy Reliable and High-Purity Piperaquine Impurities, Delivered by Synchemia Research Chemicals.
 Download catalogue
Download catalogue
Pharmaceutical Reference Standards
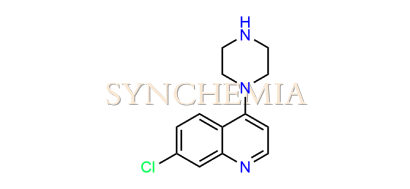
| CAT No | SRC-P16311 |
| CAS No | 837-52-5 |
| Mol.F. | C13H14ClN3 |
| Mol.Wt. | 247.7 g/mol |
| INV STATUS | IN STOCK |
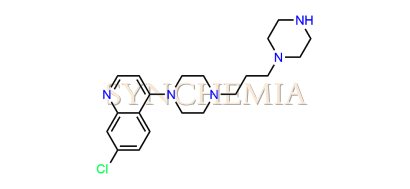
| CAT No | SRC-P16312 |
| CAS No | 4039-00-3 |
| Mol.F. | C20H28ClN5 |
| Mol.Wt. | 373.9 g/mol |
| INV STATUS | IN STOCK |
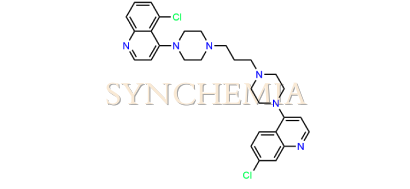
| CAT No | SRC-P16313 |
| CAS No | 918879-09-1 |
| Mol.F. | C29H32Cl2N6 |
| Mol.Wt. | 535.5 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
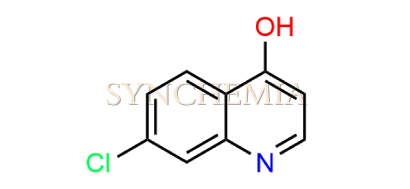
| CAT No | SRC-P16314 |
| CAS No | 86-99-7 |
| Mol.F. | C9H6ClNO |
| Mol.Wt. | 179.6 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
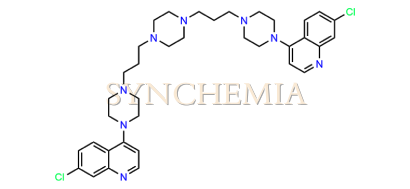
| CAT No | SRC-P16315 |
| CAS No | 86486-21-7 |
| Mol.F. | C36H46Cl2N8 |
| Mol.Wt. | 661.7 g/mol |
| INV STATUS | IN STOCK |
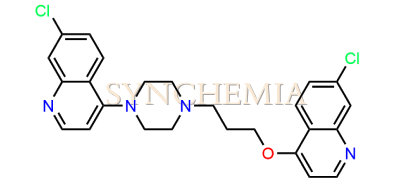
| CAT No | SRC-P16316 |
| CAS No | 1233345-93-1 |
| Mol.F. | C25H24Cl2N4O |
| Mol.Wt. | 467.4 g/mol |
| INV STATUS | IN STOCK |
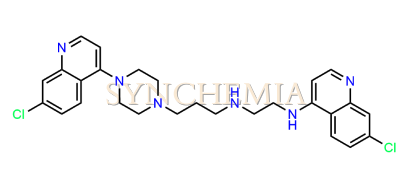
| CAT No | SRC-P16317 |
| CAS No | NA |
| Mol.F. | C27H30Cl2N6 |
| Mol.Wt. | 509.5 g/mol |
| INV STATUS | IN STOCK |
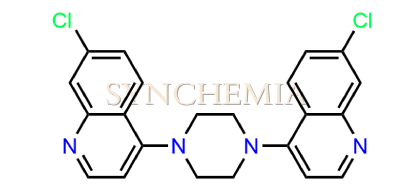
| CAT No | SRC-P16318 |
| CAS No | 31502-87-1 |
| Mol.F. | C22H18Cl2N4 |
| Mol.Wt. | 409.3 g/mol |
| INV STATUS | IN STOCK |
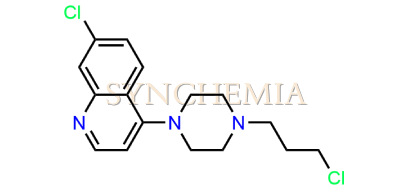
| CAT No | SRC-P16319 |
| CAS No | 4038-99-7 |
| Mol.F. | C16H19Cl2N3 |
| Mol.Wt. | 324.2 g/mol |
| INV STATUS | IN STOCK |
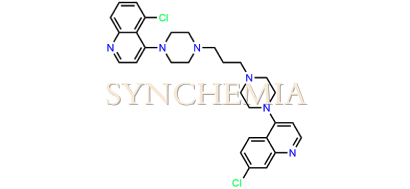
| CAT No | SRC-P16320 |
| CAS No | 918879-09-1 |
| Mol.F. | C29H32Cl2N6 |
| Mol.Wt. | 535.5 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
Let’s advance your research together.
Partner with us for specialized chemical materials and synthesis services that enable pharmaceutical innovation.
 info@synchemia.com | sales@synchemia.com
info@synchemia.com | sales@synchemia.com  9766925136
9766925136 


