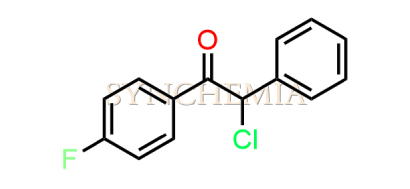
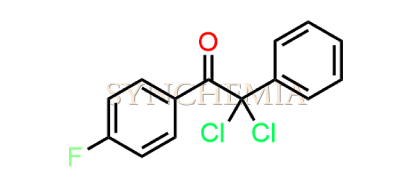
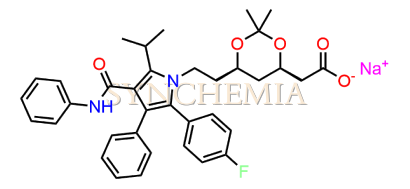
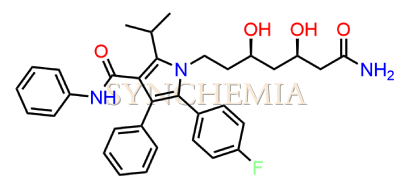
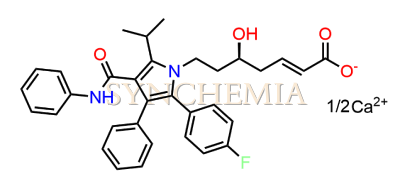
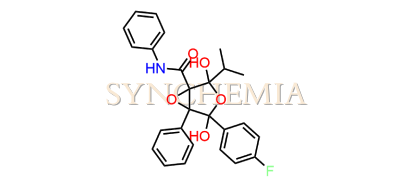
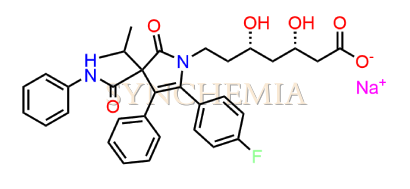
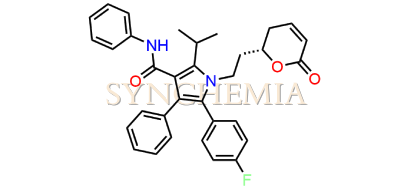
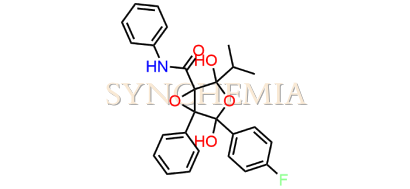
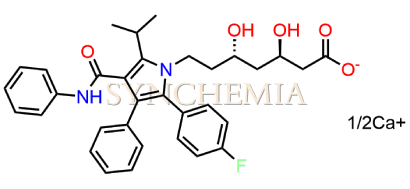
Atorvastatin EP Impurity G (Calcium Salt)

Atorvastatin EP Impurity G (Calcium Salt) | CAS NO: 887196-29-4, also known as Atorvastatin EP Impurity G its chemical | IUPAC name is (3R,5R)-7-[2-(4-fluorophenyl)-3-phenyl-4-(phenylcarbamoyl)-5-(propan-2-yl)-1H-pyrrol-1-yl]-5-hydroxy-3-methoxyheptanoic acid Calcium salt. Atorvastatin EP Impurity G (Calcium Salt) is potential impurity of Atorvastatin. It helps reduce levels of low-density lipoprotein (LDL) cholesterol and total cholesterol, which can help prevent cardiovascular disease. It’s often prescribed to lower the risk of heart attack and stroke in individuals with high cholesterol or those with risk factors for heart disease. Synchemia Research chemical is manufacturer and Supplier of Atorvastatin EP Impurity G (Calcium Salt) with comprehensive characterization data that adheres to regulatory guidelines, ensuring its suitability for pharmaceutical QC applications.
Synchemia Research chemical is a leading manufacturer, Supplier and exporter of high-quality Atorvastatin EP Impurity G (Calcium Salt) | CAS NO: 887196-29-4, also known as Atorvastatin EP Impurity G (Calcium Salt). Atorvastatin is used to treat conditions characterized by abnormal lipid levels, including hyperlipidemia and mixed dyslipidemia. It can help raise high-density lipoprotein (HDL) cholesterol levels, which is considered beneficial for heart health. Buy Reliable and High-Purity Atorvastatin EP Impurity G (Calcium Salt), Delivered by Synchemia Research Chemical.
 info@synchemia.com | sales@synchemia.com
info@synchemia.com | sales@synchemia.com  9766925136
9766925136