Rivaroxaban
Rivaroxaban | CAS NO: 366789-02-8 Rivaroxaban is an oral anticoagulant medication used to prevent and treat various conditions related to blood clots. Prevention of Stroke and Systemic Embolism: In patients with non-valvular atrial fibrillation, rivaroxaban helps reduce the risk of stroke and systemic embolism (blockage of blood flow to other parts of the body).Treatment of Deep Vein Thrombosis (DVT): It is used to treat DVT, a condition where blood clots form in the deep veins of the legs. Prevention of DVT and Pulmonary Embolism (PE) After Surgery: Rivaroxaban is used to prevent DVT and PE in patients who have undergone hip or knee replacement surgery.Treatment of Pulmonary Embolism (PE): It helps treat PE, a condition where blood clots block the arteries in the lungs. Rivaroxaban works by inhibiting Factor Xa, an essential component in the blood clotting process, which helps to prevent the formation of new clots and the growth of existing ones.
We are the Synchemia Research Chemical specializes in manufacturing and supplying of Rivaroxaban impurities that meet specific pharmacopeial standards (EP/BP/USP/IP) as well as process-related impurities Rivaroxaban works by inhibiting Factor Xa, an essential component in the blood clotting process, which helps to prevent the formation of new clots and the growth of existing ones. It is typically taken as a tablet and is often chosen for its convenience and fixed dosing regimen compared to some other anticoagulants. we the Synchemia are ensuring the purity and quality of its impurities which is crucial for pharmaceutical API manufacturing companies for regulatory compliance. Buy Reliable and High-Purity Rivaroxaban Impurities, Delivered by Synchemia Research Chemicals.
 Download catalogue
Download catalogue
Pharmaceutical Reference Standards
.png)
| CAT No | SRC-R18141 |
| CAS No | 366789-02-8 |
| Mol.F. | C19H18ClN3O5S |
| Mol.Wt. | 435.9 g/mol |
| INV STATUS | IN STOCK |
.png)
| CAT No | SRC-R18142 |
| CAS No | 865479-71-6 |
| Mol.F. | C19H18CIN3O5S |
| Mol.Wt. | 435.9 g/mol |
| INV STATUS | IN STOCK |
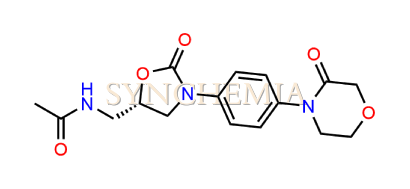
| CAT No | SRC-R18143 |
| CAS No | 1429334-00-8 |
| Mol.F. | C16H19N3O5 |
| Mol.Wt. | 333.3 g/mol |
| INV STATUS | IN STOCK |
.png)
| CAT No | SRC-R18144 |
| CAS No | 1365267-35-1 |
| Mol.F. | C29H32N6O9 |
| Mol.Wt. | 608.6 g/mol |
| INV STATUS | IN STOCK |
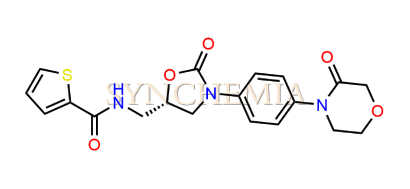
| CAT No | SRC-R18145 |
| CAS No | 1415566-28-7 |
| Mol.F. | C19H19N3O5S |
| Mol.Wt. | 401.4 g/mol |
| INV STATUS | IN STOCK |
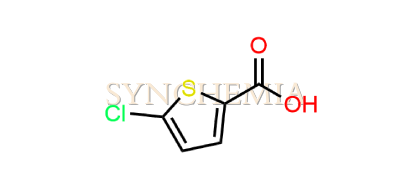
| CAT No | SRC-R18146 |
| CAS No | 24065-33-6 |
| Mol.F. | C5H3ClO2S |
| Mol.Wt. | 162.6 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |

| CAT No | SRC-R18147 |
| CAS No | 446292-08-6 |
| Mol.F. | C22H19N3O6 |
| Mol.Wt. | 421.4 g/mol |
| INV STATUS | IN STOCK |
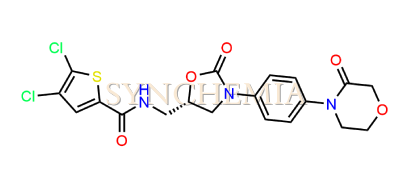
| CAT No | SRC-R18148 |
| CAS No | 1770812-37-7 |
| Mol.F. | C19H17Cl2N3O5S |
| Mol.Wt. | 470.3 g/mol |
| INV STATUS | IN STOCK |
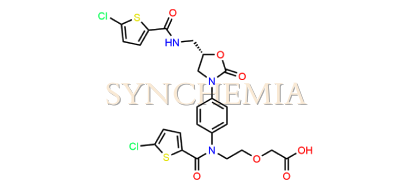
| CAT No | SRC-R18149 |
| CAS No | 1151893-81-0 |
| Mol.F. | C24H21Cl2N3O7S2 |
| Mol.Wt. | 598.5 g/mol |
| INV STATUS | IN STOCK |
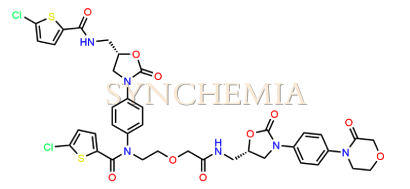
| CAT No | SRC-R18150 |
| CAS No | 1632463-24-1 |
| Mol.F. | C38H36CI2N6O10S2 |
| Mol.Wt. | 871.8 g/mol |
| INV STATUS | IN STOCK |
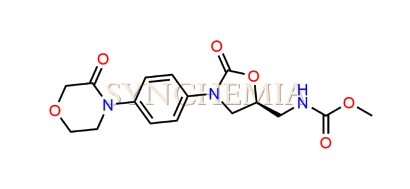
| CAT No | SRC-R18151 |
| CAS No | NA |
| Mol.F. | C16H19N3O6 |
| Mol.Wt. | 349.3 g/mol |
| INV STATUS | IN STOCK |

| CAT No | SRC-R18152 |
| CAS No | 1838139-24-4 |
| Mol.F. | C19H18ClN3O5S |
| Mol.Wt. | 435.9 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
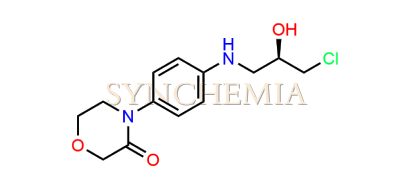
| CAT No | SRC-R18153 |
| CAS No | 1252018-10-2 |
| Mol.F. | C13H17ClN2O3 |
| Mol.Wt. | 284.7 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
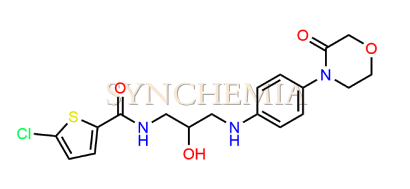
| CAT No | SRC-R18154 |
| CAS No | 482305-98-6 |
| Mol.F. | C18H20N3O4ClS |
| Mol.Wt. | 409.9 g/mol |
| INV STATUS | IN STOCK |
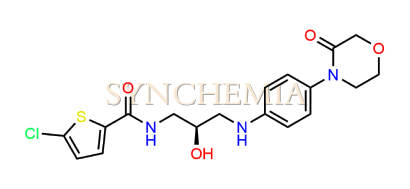
| CAT No | SRC-R18155 |
| CAS No | 721401-53-2 |
| Mol.F. | C18H20CIN3O4S |
| Mol.Wt. | 409.9 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
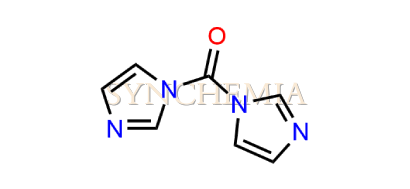
| CAT No | SRC-R18156 |
| CAS No | 530-62-1 |
| Mol.F. | C7H6N4O |
| Mol.Wt. | 162.2 g/mol |
| INV STATUS | CUSTOM SYNTHESIS |
Let’s advance your research together.
Partner with us for specialized chemical materials and synthesis services that enable pharmaceutical innovation.
 info@synchemia.com | sales@synchemia.com
info@synchemia.com | sales@synchemia.com  9766925136
9766925136 



