- A
- B
- C
- D
- E
- F
- G
- H
- I
- J
- K
- L
- M
- N
- O
- P
- Q
- R
- S
- T
- U
- V
- W
- X
- Y
- Z
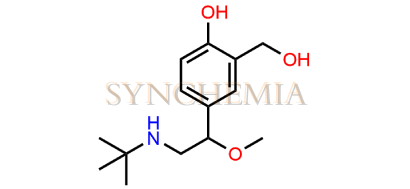
Salbutamol EP Impurity A
Synchemia is a leading supplier of pharmaceutical-grade Salbutamol impurities, offering high-quality reference impurity standards for research and analytical purposes. Salbutamol EP Impurity A Impurity A is a specified degradation product or synthetic intermediate in the manufacture of salbutamol. It is controlled under EP monographs to ensure drug safety, efficacy, and purity. Excess levels can indicate instability, poor manufacturing control, or storage degradation.
Salbutamol EP Impurity A | CAS NO: 870076-72-5, also known as Salbutamol EP Impurity A is chemically expressed as 4-[(1RS)-2-(tert-butylamino)-1-methoxyethyl]-2-(hydroxymethyl)phenol (as per EP). Salbutamol EP Impurity A is supplied with comprehensive characterization data, in full compliance with applicable regulatory guidelines. Synchemia is a leading supplier and exporter of Salbutamol EP Impurity A. All Synchemia products are strictly intended for analytical testing purposes only and not for human use.
| Product name | Salbutamol EP Impurity A |
| SRC CAT No | SRC-S19142 |
| CAS No | 870076-72-5 |
| Mol.F. | C14H23NO3 |
| Mol.Wt. | 253.3 g/mol |
| Related CAS No. | 2468639-65-6 (HCl salt) |
| Inv Status | Custom Synthesis |