- A
- B
- C
- D
- E
- F
- G
- H
- I
- J
- K
- L
- M
- N
- O
- P
- Q
- R
- S
- T
- U
- V
- W
- X
- Y
- Z
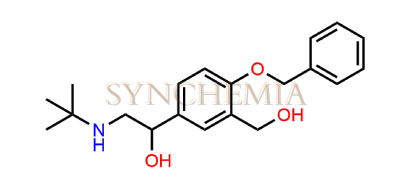
Salbutamol EP Impurity I
Synchemia is a leading supplier of pharmaceutical-grade Salbutamol impurities, offering high-quality reference impurity standards for research and analytical purposes. Salbutamol EP Impurity I Used as a reference impurity standard for method development, validation, and quality control. It helps in ensuring that analytical methods (e.g. HPLC) can separate and quantify this impurity.
Salbutamol EP Impurity I | CAS NO: 56796-66-8, also known as Salbutamol BP Impurity I ; 4-Benzyl Albuterol is chemically expressed as (1RS)-1-[4-(benzyloxy)-3-(hydroxymethyl)phenyl]-2-(tert-butylamino)ethan-1-ol. Salbutamol EP Impurity I is supplied with comprehensive characterization data, in full compliance with applicable regulatory guidelines. Synchemia is a leading supplier and exporter of Salbutamol EP Impurity I. All Synchemia products are strictly intended for analytical testing purposes only and not for human use.
| Product name | Salbutamol EP Impurity I |
| SRC CAT No | SRC-S19150 |
| CAS No | 56796-66-8 |
| Mol.F. | C20H27NO3 |
| Mol.Wt. | 329.4 g/mol |
| Related CAS No. | NA |
| Inv Status | Custom Synthesis |