- A
- B
- C
- D
- E
- F
- G
- H
- I
- J
- K
- L
- M
- N
- O
- P
- Q
- R
- S
- T
- U
- V
- W
- X
- Y
- Z
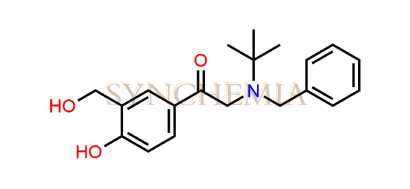
Salbutamol EP Impurity G
Synchemia is a leading supplier of pharmaceutical-grade Salbutamol impurities, offering high-quality reference impurity standards for research and analytical purposes. Salbutamol EP Impurity G mpurity G is a specified impurity listed in the European Pharmacopoeia (EP) for salbutamol. It arises likely as a process‑related impurity during the synthetic steps or due to side reactions. Because of its structural relation to salbutamol, and a benzyl substituent, it's somewhat more complex than simpler degradation products. The presence of the benzyl(tert‑butyl)amino moiety makes it a modification of the typical amine substituent.
Salbutamol EP Impurity G | CAS NO: 64092-10-0, also known as Salbutamol BP Impurity G ; N-Benzyl Salbutamone HCl ; Benzylsalbutamon is chemically expressed as 2-[Benzyl(tert-butyl)amino]-1-[4-hydroxy-3-(hydroxymethyl)phenyl]ethan-1-one(as per EP). Salbutamol EP Impurity G is supplied with comprehensive characterization data, in full compliance with applicable regulatory guidelines. Synchemia is a leading supplier and exporter of Salbutamol EP Impurity G. All Synchemia products are strictly intended for analytical testing purposes only and not for human use.
| Product name | Salbutamol EP Impurity G |
| SRC CAT No | SRC-S19148 |
| CAS No | 64092-10-0 |
| Mol.F. | C20H25NO3 |
| Mol.Wt. | 327.4 g/mol |
| Related CAS No. | NA |
| Inv Status | Custom Synthesis |